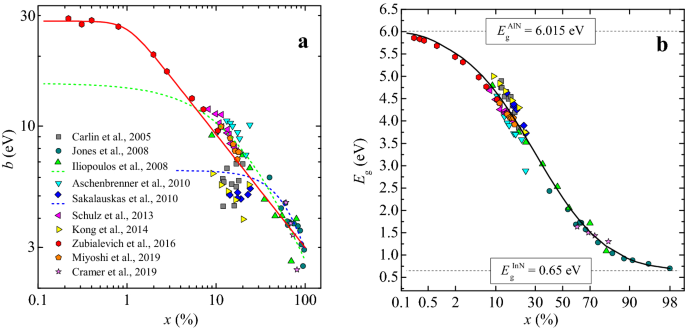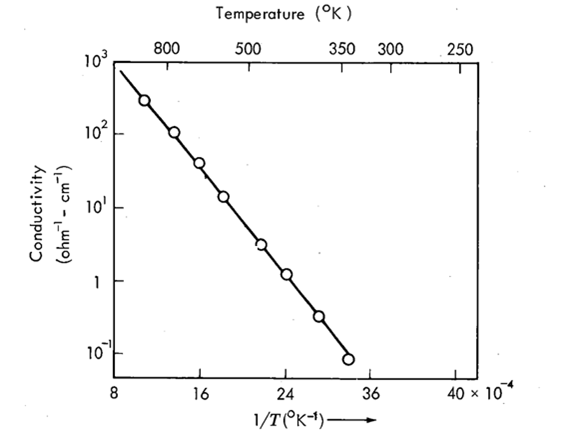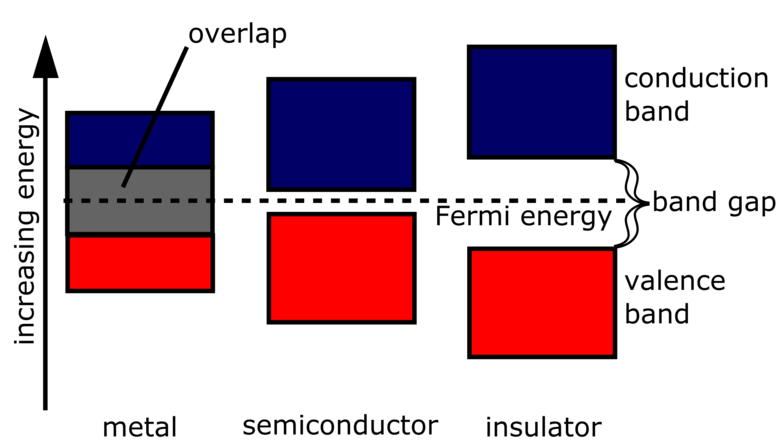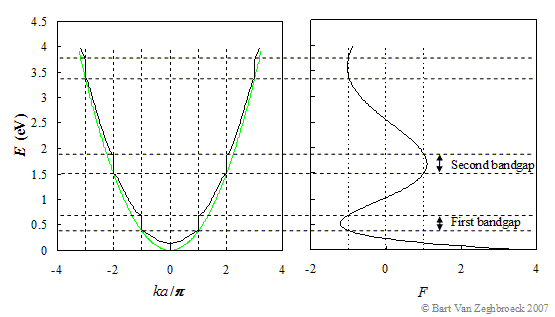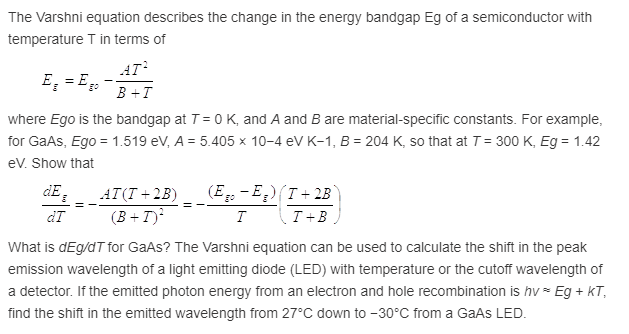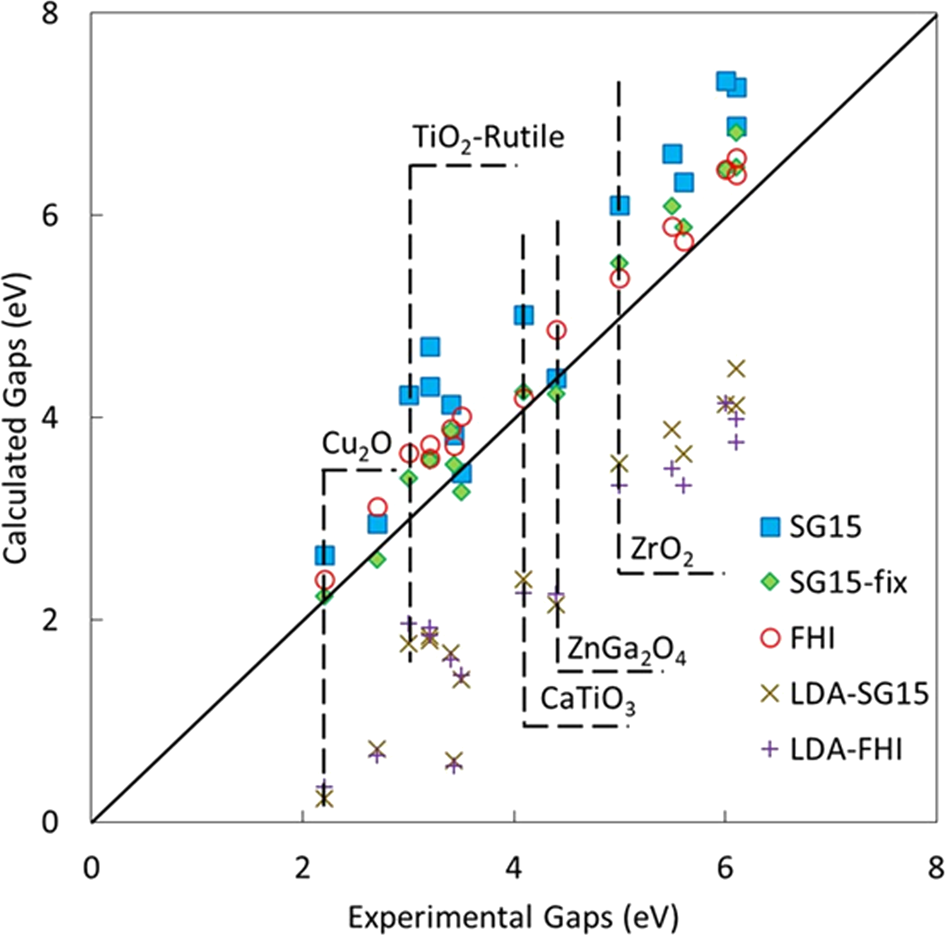
Wannier–Koopmans method calculations for transition metal oxide band gaps | npj Computational Materials

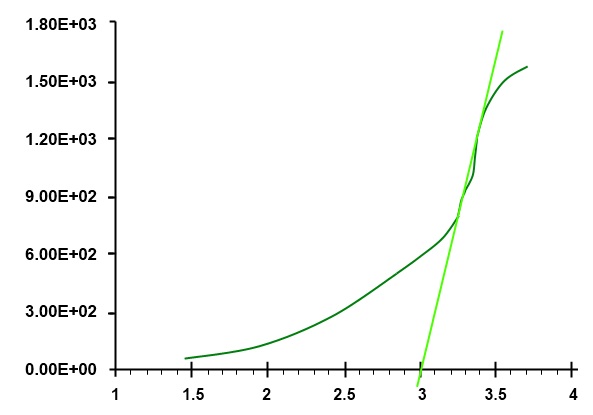
How To Correctly Determine the Band Gap Energy of Modified Semiconductor Photocatalysts Based on UV–Vis Spectra | The Journal of Physical Chemistry Letters

How To Correctly Determine the Band Gap Energy of Modified Semiconductor Photocatalysts Based on UV–Vis Spectra | The Journal of Physical Chemistry Letters

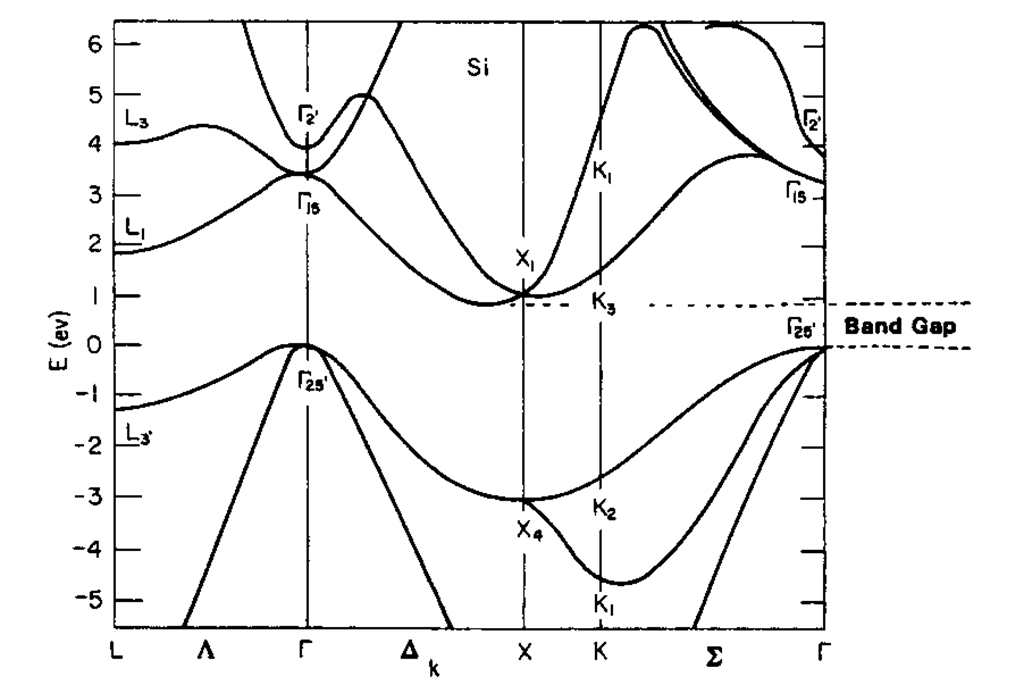
If an LED has to emit 662 nm wavelength of light them what should be the band gap energy of its semiconductor? h = 6.62 × 10^-34 Js.